What Is the Fetal PR Interval?
The PR interval — the time from atrial depolarisation to ventricular depolarisation — is one of cardiology’s most fundamental measurements. In the fetus, we cannot record it directly. There is no practical bedside ECG. What pulsed-wave Doppler gives us instead is a mechanical surrogate: the time between the onset of atrial contraction and the onset of ventricular ejection.
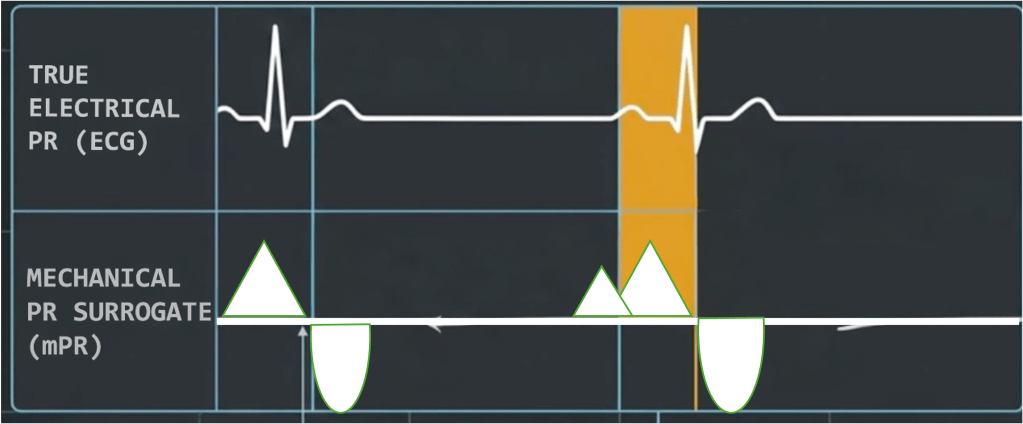
Figure 1. The relationship between the true electrical PR interval (ECG) and the mechanical PR surrogate (mPR) measured by pulsed-wave Doppler. The mPR consistently exceeds the electrical PR by approximately 14–16 ms. This is a systematic, predictable difference and not random error, and must be kept in mind when interpreting Doppler-derived values against clinical thresholds.
This is not a flaw unique to one technique. Every standard pulsed-wave method shares it. The mPR is always longer than the true electrical PR by approximately 14–16 ms, and this is a systematic, predictable bias rather than random error.
Why Acquisition Precision Matters
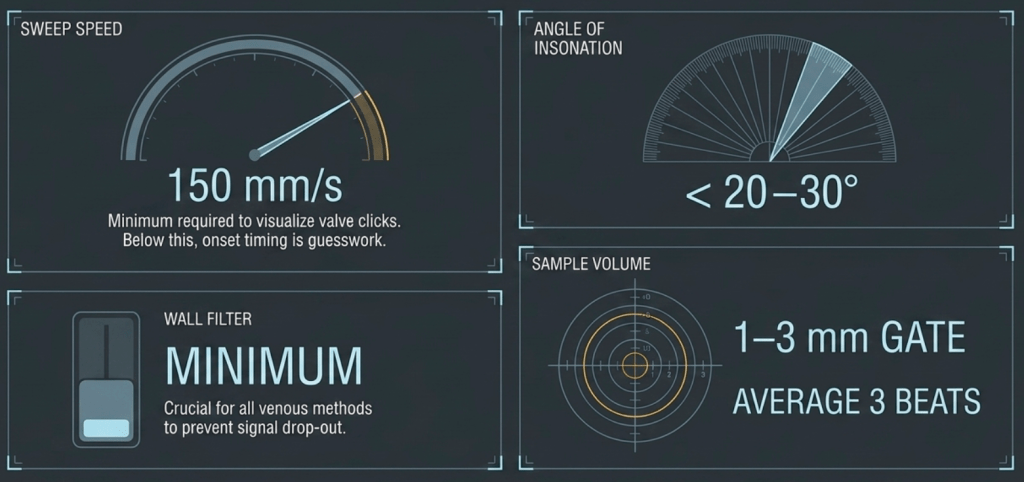
Figure 2. At a clinical treatment threshold of 150 ms, a 10–15 ms acquisition error is not a technical footnote. It dictates treatment.
The fetal mPR sits at the intersection of technique and consequence. A 15 ms error from poor sweep speed, misplaced sample volume, or the wrong fetal position is not recoverable at the reporting stage. It is built into the number.
The Three Methods
Not every fetal position allows every method. The choice is dictated by what the fetus gives you.
LV Inflow / Outflow (MV/Ao) — The Default
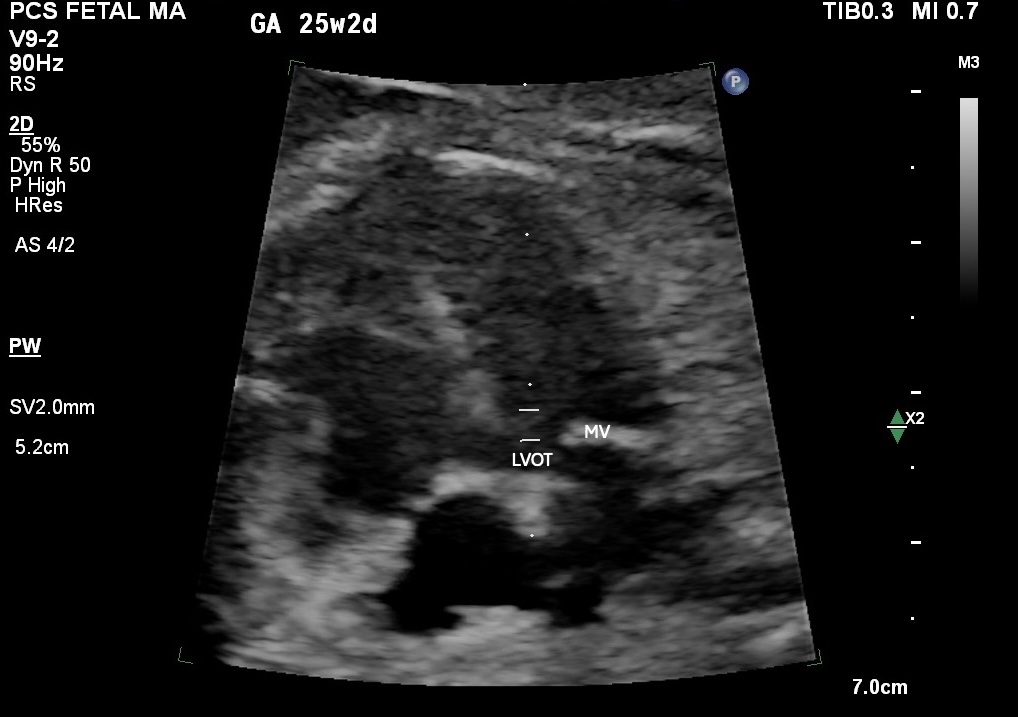
Figure 3. Sample volume positioning at the anterior mitral leaflet hinge point within the LVOT, 5-chamber view. GA 25w2d. The gate must simultaneously capture mitral inflow and aortic outflow. Not deeper into the LVOT, and not at the leaflet tips.
This method requires apex-up or apex-down fetal orientation, a sweep speed of at least 150 mm/s, and an angle of insonation below 20 degrees. The sample volume goes at the anterior mitral leaflet hinge point within the LVOT.
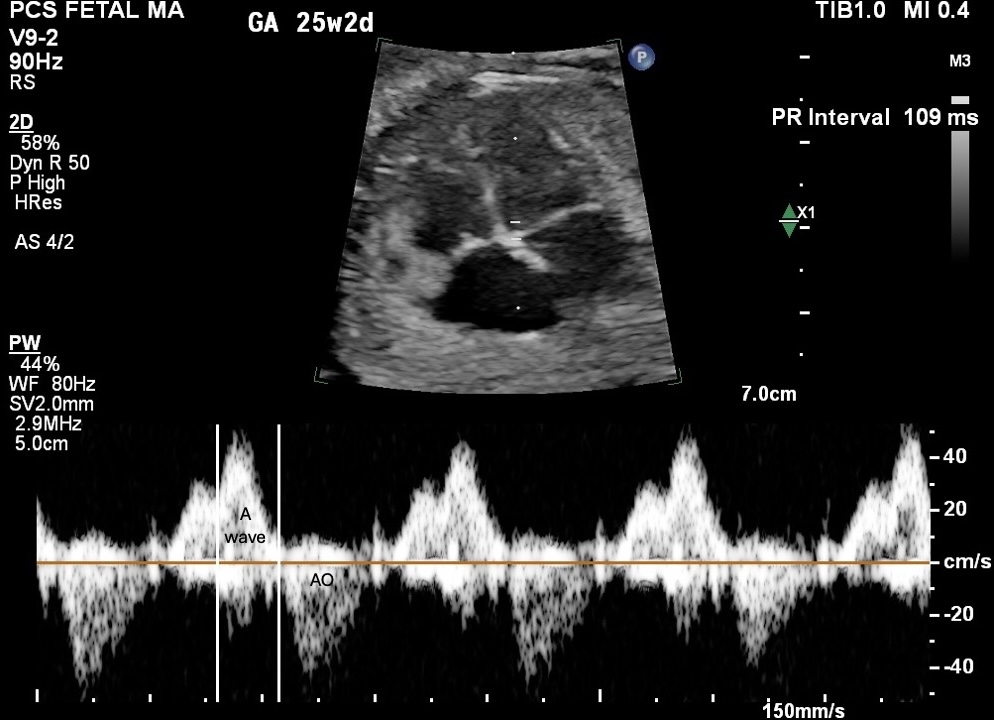
Figure 4. LV inflow/outflow Doppler trace. A-wave above baseline (atrial contraction) and aortic ejection below. PR Interval 109 ms. GA 25w2d, 150 mm/s. Measurement from the onset of the A-wave to the onset of aortic ejection.
Measure from the foot of the A-wave to the leading edge of the aortic opening click. Average three beats minimum. If E and A waves are fused, which occurs at heart rates above approximately 160 bpm, switch to SVC/Ao.
SVC / Ascending Aorta (SVC/Ao) — The Rescue
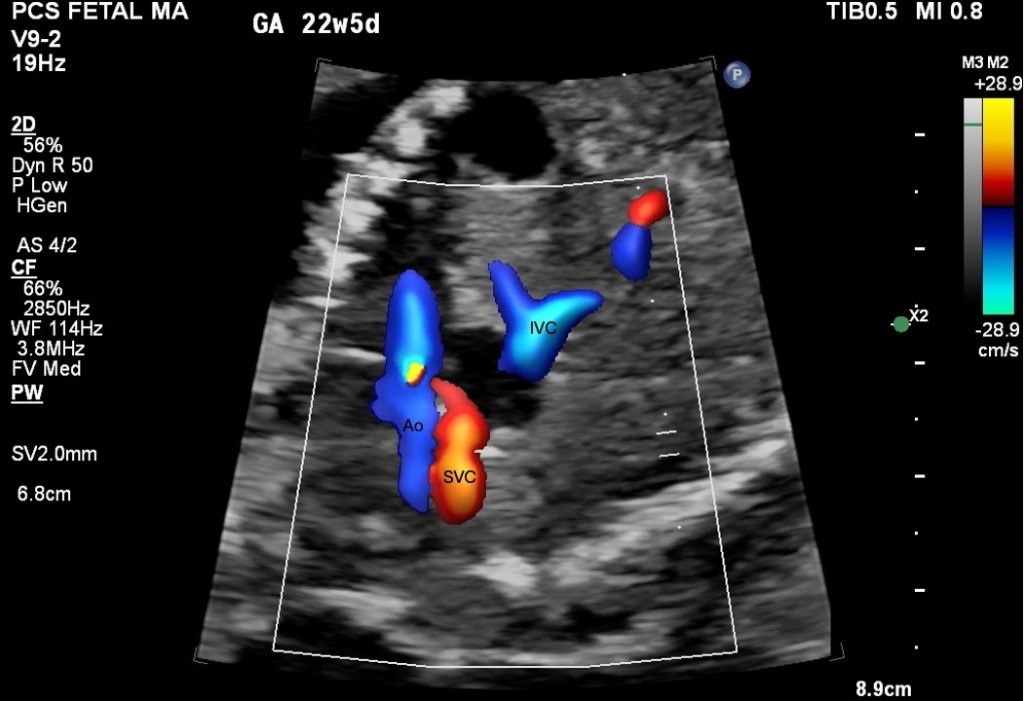
Figure 5. Colour Doppler showing the SVC and ascending aorta (Ao) with IVC visible. GA 22w5d. The SVC runs parallel to the Ao over a short mediastinal segment, allowing simultaneous sampling.
This method requires a near-zero angle of insonation. The wall filter must be set to minimum before sampling. The SVC a-wave reversal is a low-velocity signal, and the default wall filter will suppress it entirely. If the a-wave is absent from your trace, check the filter before repositioning the probe.
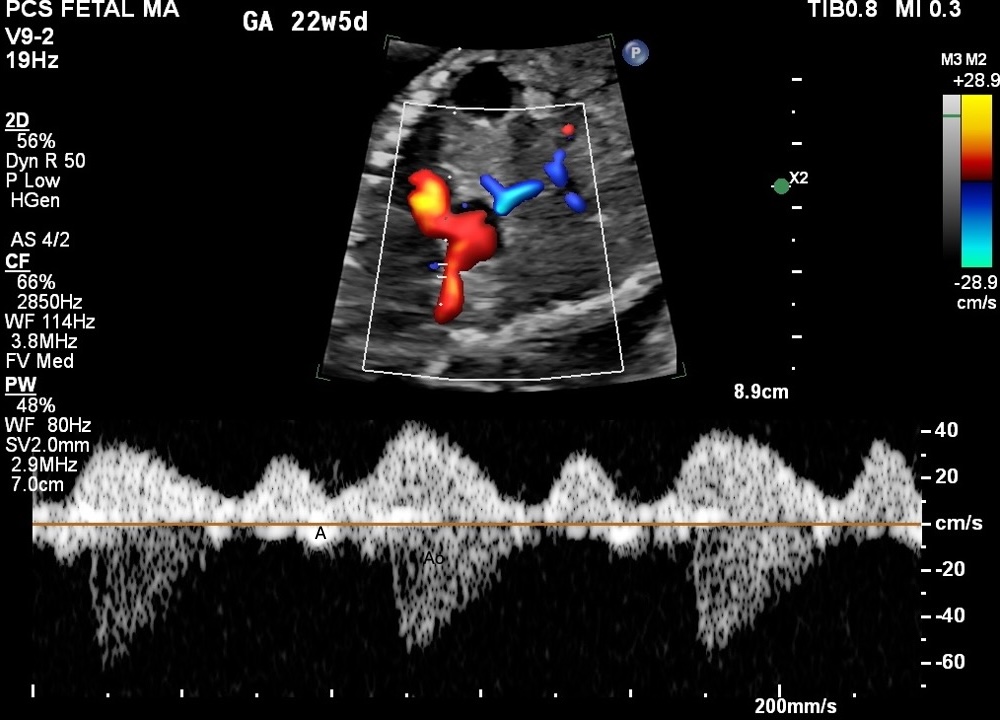
Figure 6. SVC/Ao Doppler trace. SVC a-wave reversal (A, above baseline) marks atrial systole. Aortic ejection (Ao, below baseline) marks ventricular ejection. GA 22w5d.
SVC/Ao gives broadly similar values to MV/Ao in fetal studies. Kato et al. (2012) showed virtually identical overestimation of the electrical PR for both methods (14.6% vs 14.7%). Switching to SVC/Ao as a rescue method does not introduce a clinically meaningful numerical offset.
Pulmonary Artery / Vein (PA/PV) — The Fallback
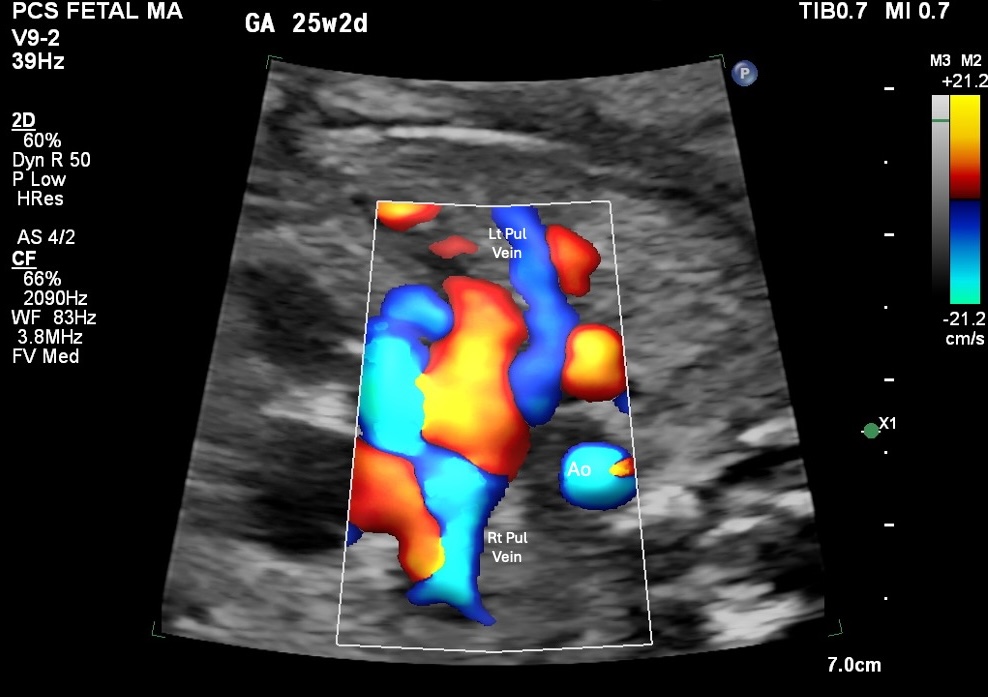
Figure 7. Colour Doppler showing left and right pulmonary veins and aorta (Ao). GA 25w2d. The right pulmonary artery and an ipsilateral pulmonary vein are the target vessels for simultaneous sampling.
The PA/PV method is position-independent, which is its main advantage when both MV/Ao and SVC/Ao are limited by fetal position. One critical difference to understand: the pulmonary venous atrial signal is a notch or velocity reduction in forward flow, not a retrograde reversal as seen in the SVC.
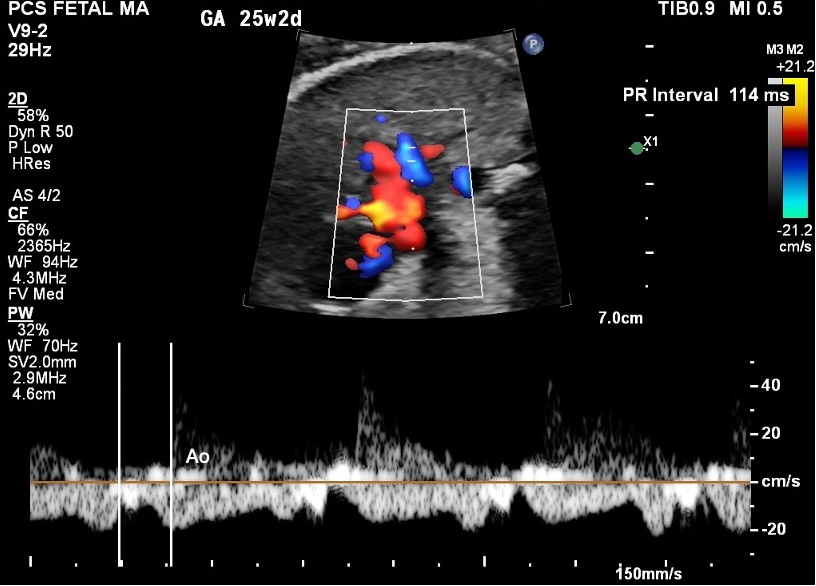
Figure 8. PA/PV Doppler trace showing pulmonary artery flow. PR 114 ms. GA 25w2d, 150 mm/s.
Tissue Doppler Imaging (TDI) — Advanced
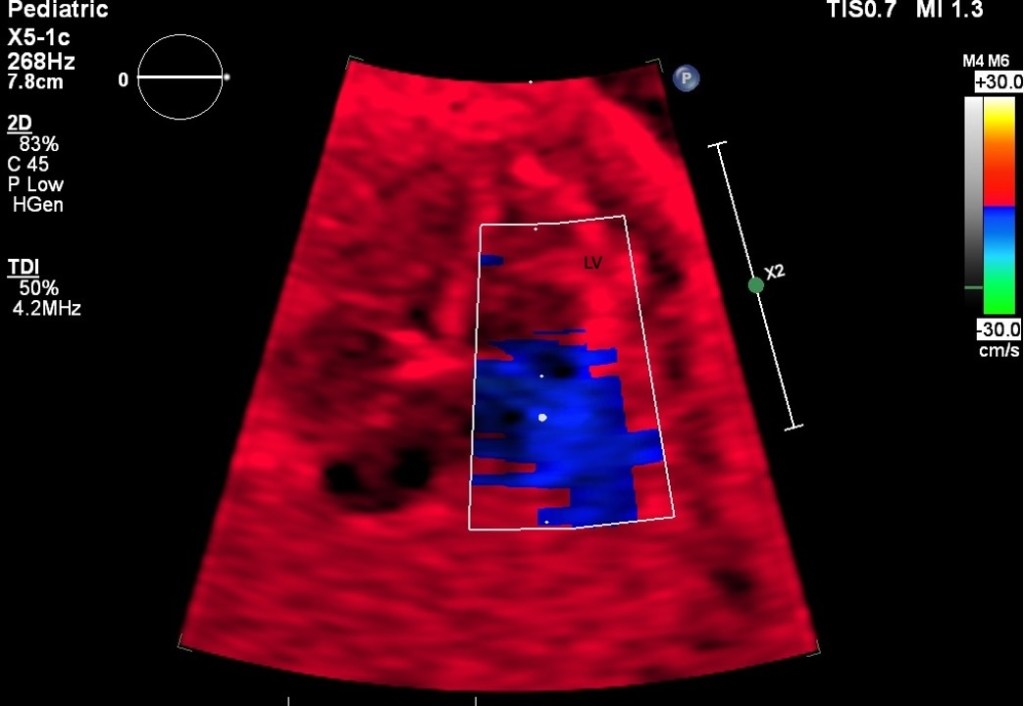
Figure 9. Colour TDI apical view showing the left ventricle (LV). This method requires true apical orientation, with the cardiac long axis running parallel to the beam.
TDI is not a first-line PR surveillance tool. It is best used when rhythm analysis rather than PR number is the primary question, or as a supplementary technique in complex arrhythmia assessment.
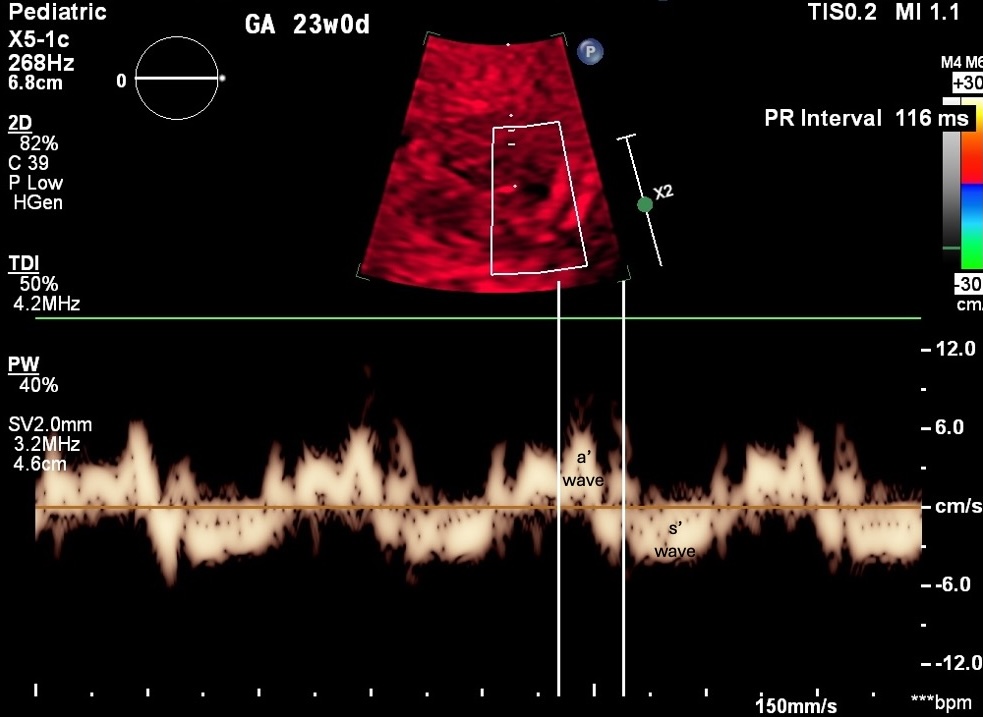
Figure 10. TDI Doppler trace. a ‘ wave marks atrial wall motion (atrial contraction surrogate). s’ wave marks ventricular systole. PR 116 ms. GA 23w0d, 150 mm/s.
Before You Probe
Figure 11. Machine settings for mPR acquisition. These are prerequisites, not preferences.
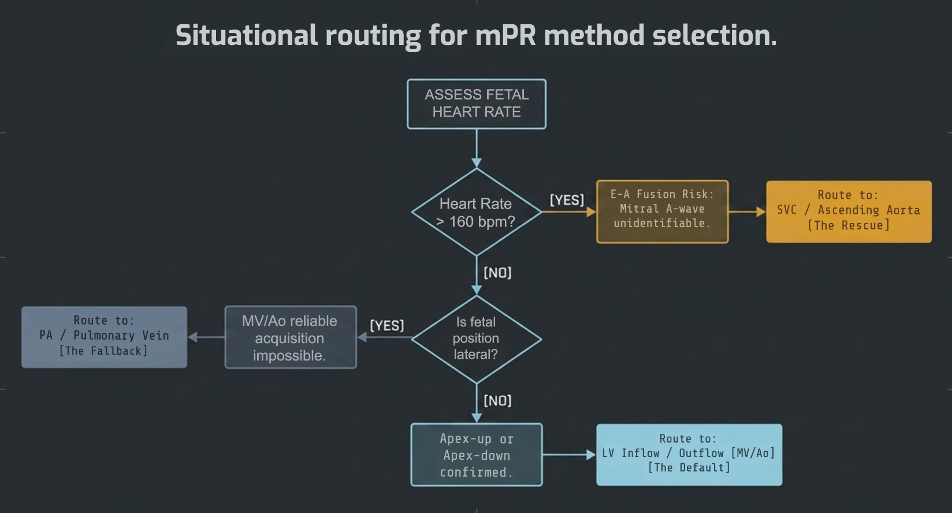
Choosing Your Method
Figure 12. Situational routing for method selection. Start by assessing fetal heart rate and fetal position. The method follows from there.
The Precision Problem

Figure 13. The artefact trap. Because PA/PV values run 6–15 ms higher than MV/Ao across gestation, switching methods mid-surveillance without accounting for this offset creates the illusion of PR prolongation where none exists.
Document your method at every study. Never compare values from different methods across serial studies without accounting for their systematic differences.
The normal values by method and gestational age, and their clinical implications, are covered in detail in the interactive guide.
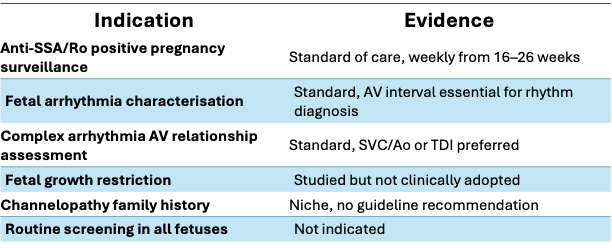
When Is mPR Measurement Indicated?
Routine mPR measurement in all fetal echos is not recommended by any current guideline. Its role is specific.
The specificity of the indication is precisely what makes acquisition precision matter. In anti-Ro positive surveillance, a 10–15 ms error at the probe is a potential treatment decision.
Clinical implications, thresholds, and what to do with the number you obtain are covered in Part 2.
REFERENCES
- Anuwutnavin S et al. Prenat Diagn 2018;38:459–466
- Bergman G et al. Ultrasound Obstet Gynecol 2006;28:57–62
- Fouron JC et al. Heart 2003;89:1211–1216
- Carvalho JS et al. Heart 2007;93:1448–1453
- Kato Y et al. Fetal Diagn Ther 2012;32:109–115
- Phoon CKL et al. Congenit Heart Dis 2012;7:349–360
- Sonesson SE. Acta Obstet Gynecol Scand 2016;95:697–704
- Swiercz G et al. J Clin Med 2025;14:7522
- Wojakowski A et al. Ultrasound Obstet Gynecol 2009;34:538–542
- Nii M et al. Heart 2006;92:1831–1837
- ASE Fetal Echocardiography Guidelines. J Am Soc Echocardiogr 2023