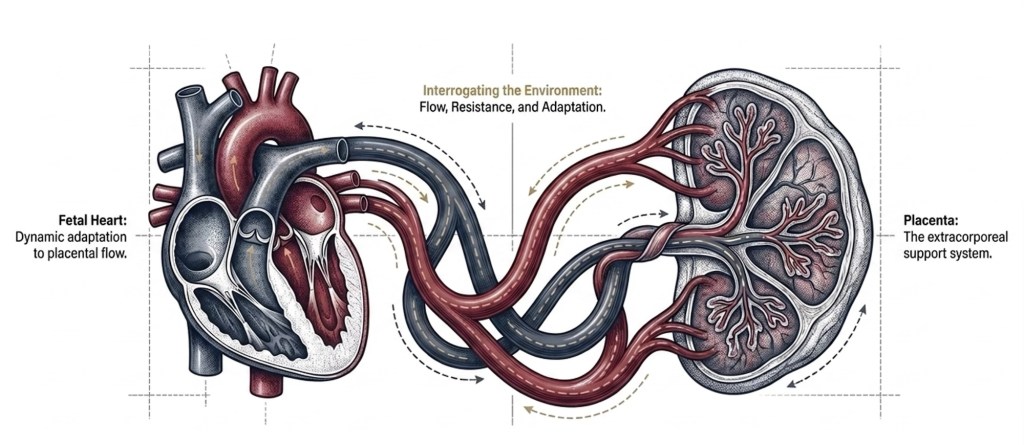
Looking Beyond the Anatomy
Fetal cardiology has historically been a structural discipline. This framework is necessary. It is also incomplete. A CHD fetus exists within a stressed biological system shaped by a placenta that is often compromised from the beginning — not as a consequence of the cardiac defect, but as a co-product of the same early injury.
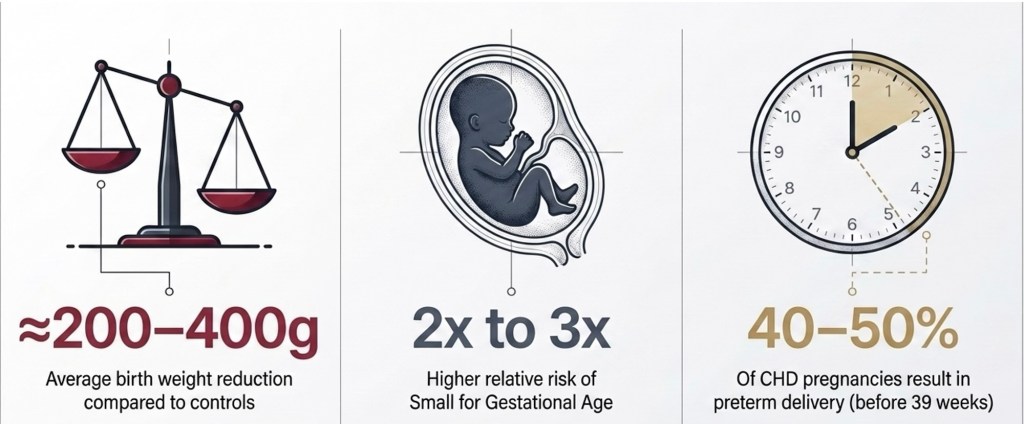
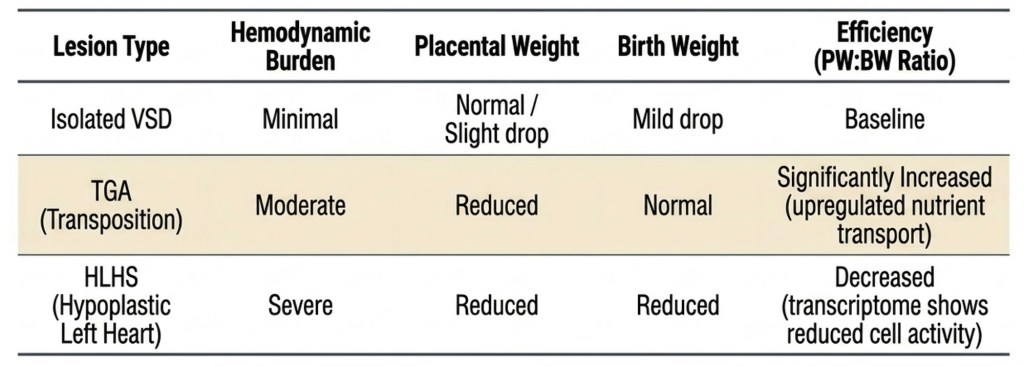
These findings persist after excluding aneuploidy and extracardiac anomalies. A fetus with an isolated VSD — zero haemodynamic compromise in utero — still shows a measurable birth weight drop. Something else is operating before the blood starts flowing.
Note: The ≈200–400g range reflects individual study estimates. The Aliasi et al. meta-analysis (BJOG 2022; 27,893 cases) reports a pooled birthweight z-score of −0.20 and a 1.6-fold higher SGA risk across all CHD types — the most rigorous summary estimate currently available.
The Preeclampsia Connection
Boyd et al. (Circulation 2017; N = 1,972,857) showed a sevenfold increased risk of early preterm preeclampsia in pregnancies with offspring CHD. The gradient — severe placental failure most strongly linked, gestational hypertension not at all — is a specific mechanistic signal, not statistical noise.
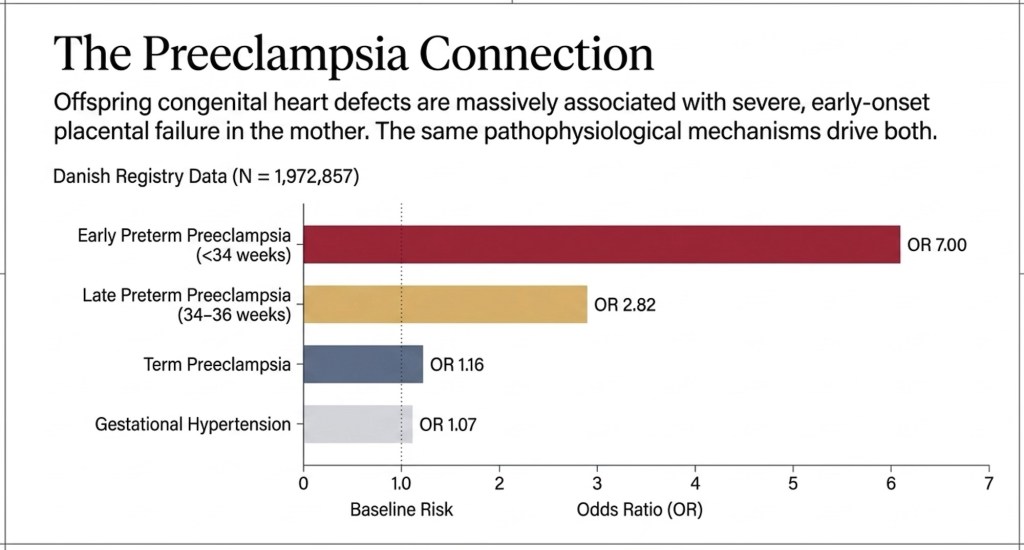
The association held across all CHD subtypes — VSD, ASD, conotruncal, left and right heart obstruction alike — pointing to a shared process operating before individual cardiac structures have formed.
Cross-Pregnancy Data: Proving a Maternal Origin
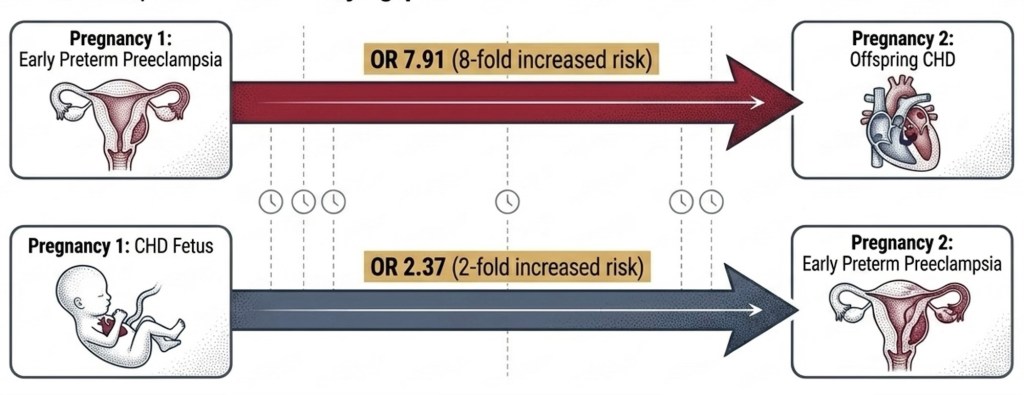
If CHD-associated preeclampsia were caused solely by the abnormal fetal heart, the risk would not carry over into a structurally normal subsequent pregnancy. It does. This points to a persistent maternal endothelial mechanism — something inherent to the mother’s vascular biology that independently raises the risk of both.
Practical implication: a history of early preterm preeclampsia in a prior pregnancy is a relevant risk factor warranting detailed fetal cardiac surveillance — not merely an obstetric footnote.
Two Phases of the Cardio-Placental Relationship
The relationship operates in two mechanistically distinct phases before converging into the same clinical problem
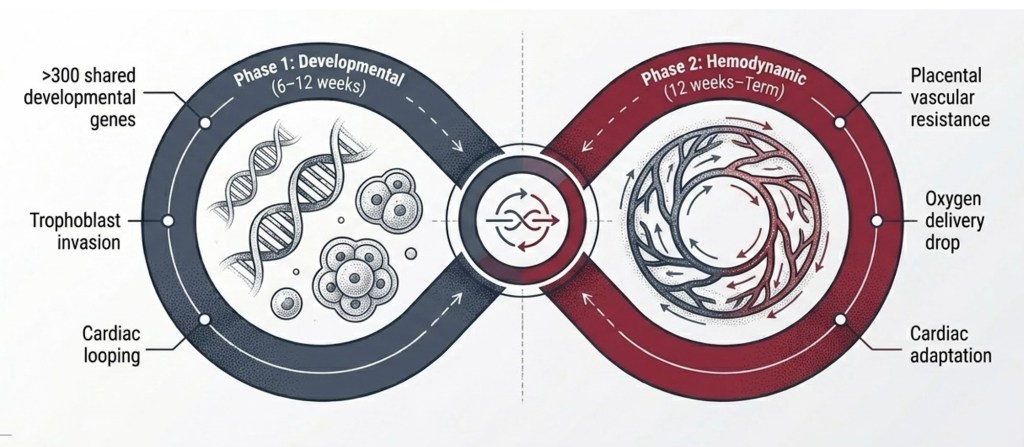
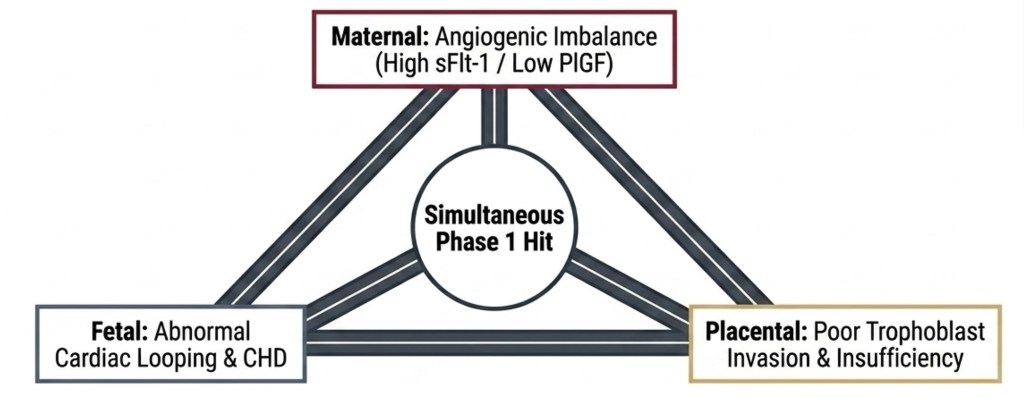
The angiogenic imbalance — elevated sFlt-1 and soluble endoglin, reduced PlGF — is measurable in maternal and cord blood in CHD pregnancies. This is not a consequence of the defect; it is part of the same Phase 1 hit.
The Phase 2 Feedback Loop
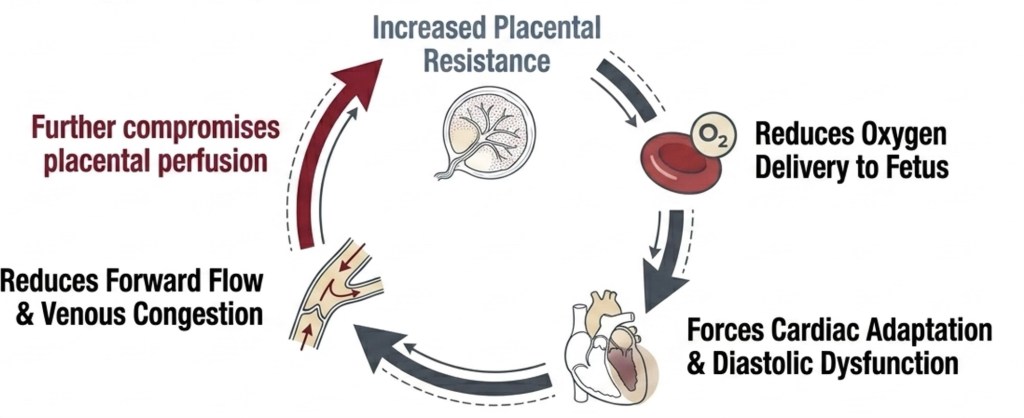
Once the circulation is established, heart and placenta trap each other. The clinical question is not whether this loop exists — it does — but when compensation fails.
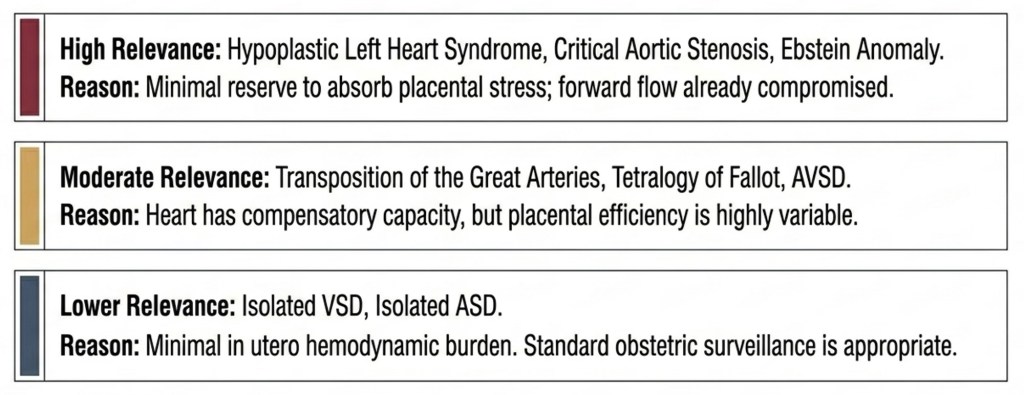
Not All Placentas Adapt Equally
Anatomy does not dictate growth outcome. Surveillance intensity must be calibrated to placental physiology, not surgical complexity.
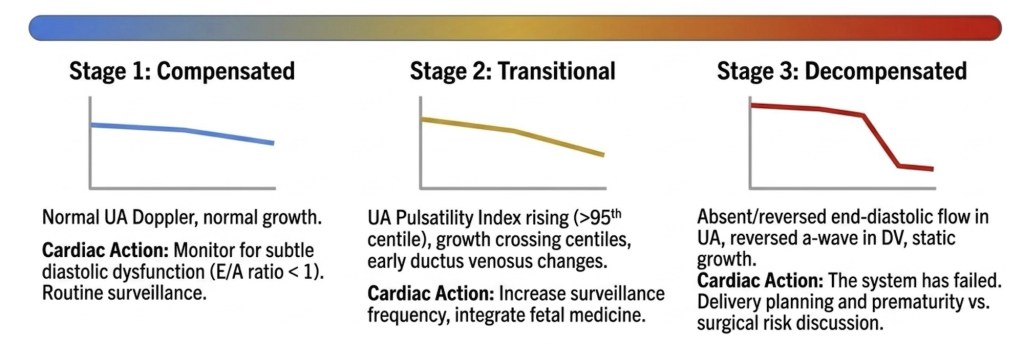
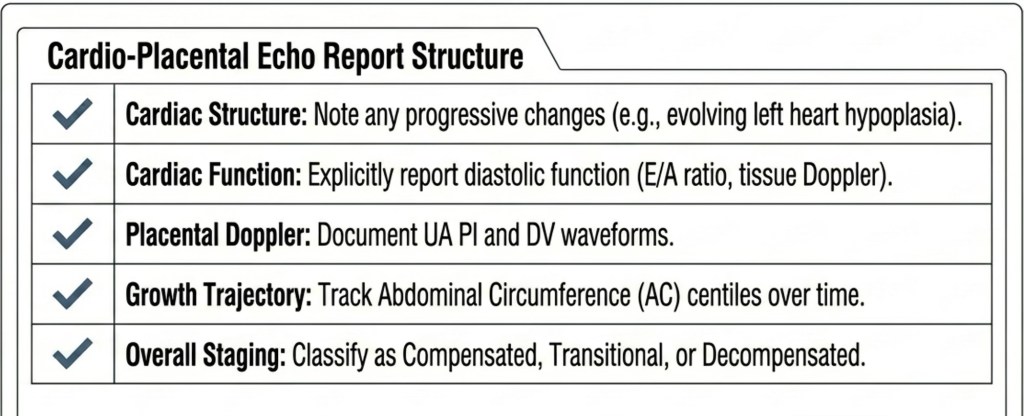
A Clinical Staging Framework
Serial cardio-placental assessment can be summarised as a clinical stage — transforming the fetal cardiology report from a description into an actionable conclusion.
Risk Stratification by Haemodynamic Burden
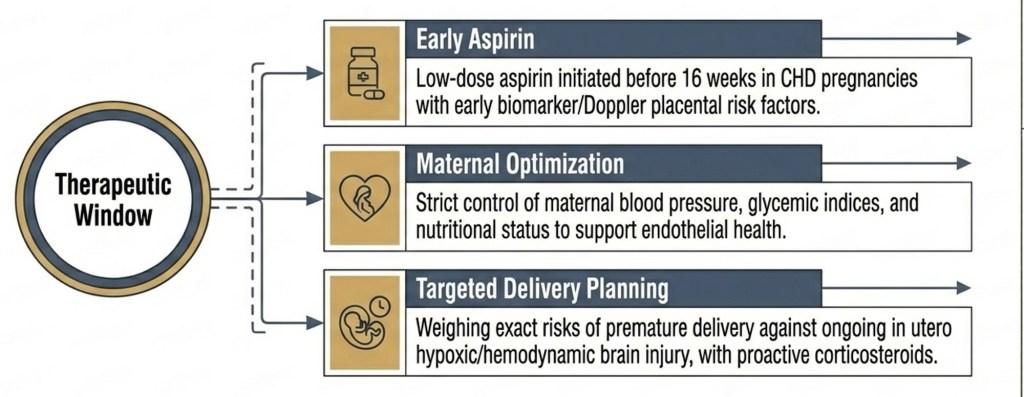
Caring for the Environment: Maternal Recommendations
If CHD and placental insufficiency share a common biological origin in maternal endothelial dysfunction, then maternal health is a modifiable determinant of intrauterine fetal wellbeing — not a background variable.
The table below addresses the remaining components of a comprehensive maternal approach.
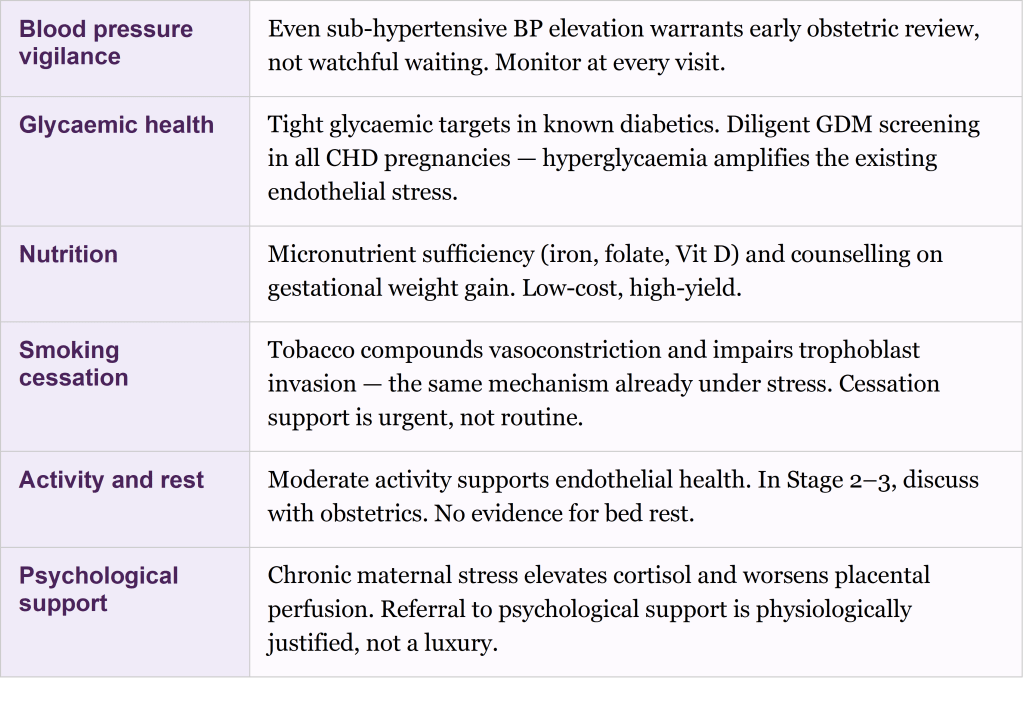
These recommendations require joint ownership between the fetal cardiologist and the obstetric team. Communicating placental risk and enquiring about maternal health optimization, is part of the modern fetal cardiology consultation, not outside it.
The Argument in One Sentence
A CHD fetus is not simply an abnormal heart waiting for postnatal surgery. It is a complex organism experiencing systemic stress, shaped by a compromised placenta and a maternal vascular environment under strain. By interrogating both, and acting on what we find, we can protect the fetal brain, extend the gestation, and redefine what fetal cardiac care means in practice
References
1. Boyd HA, et al. Association Between Fetal CHD and Maternal Risk of Hypertensive Disorders. Circulation. 2017;136:39-48.
2. Llurba E, et al. Maternal and foetal angiogenic imbalance in congenital heart defects. Eur Heart J. 2014;35:701-707.
3. Ruiz A, et al. Placenta-related complications in women carrying a foetus with CHD. J Matern Fetal Neonatal Med. 2016;29:3271-3275.
4. Jones HN, et al. HLHS is associated with structural and vascular placental abnormalities. Placenta. 2015;36:1078-1086.
5. Aliasi M, et al. Birthweight and isolated congenital heart defects: systematic review and meta-analysis. BJOG. 2022;129:1805-1816.
6. Sliwa K, Mebazaa A. Possible joint pathways of early pre-eclampsia and CHD. Eur Heart J. 2014;35:680-682.